|
Water chemistry worries Sadly, some of our TIC teachers (and I) are losing sleep over water chemistry challenges. A good example is what's going on at Mt. Anthony Union Middle School, where Emily Hunter has struggled for many days to bring down high nitrite and nitrate levels. Starting about five days ago, Emily's nitrite hit 3 ppm and her nitrate reached 80 ppm. Over the next few days, Emily did several water changes, and her nitrate dropped some, reaching 20 ppm today, but nitrite remained high, even registering 5 ppm a couple of days ago. So what can we advise Emily?
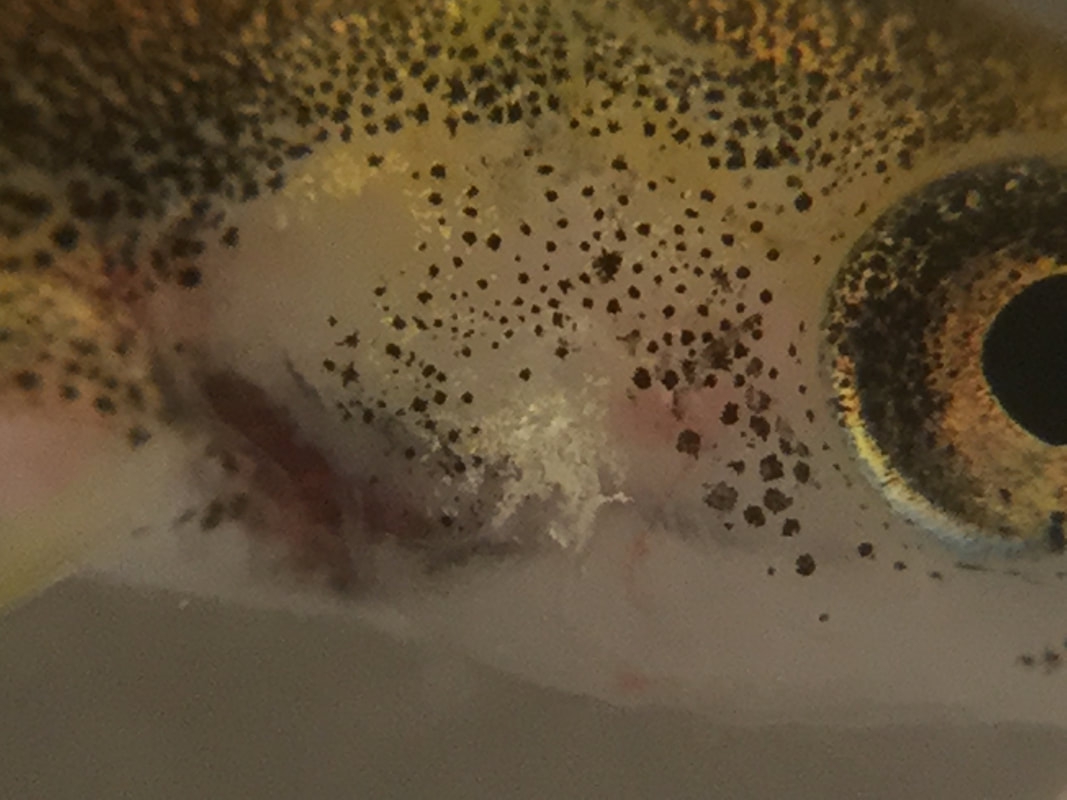
Dropping the basket at SLC Here's a video that Danielle Levine, of Schoolhouse Learning Center, sent me of the day she and her students lowered the breeder basket and released their fry into the wide world of their 55-gallon tank. Danielle also provided these photos of her liberated fish. Thanks for those submissions, Danielle! Fishy infections and other problems Along with water chemistry challenges, some of our schools have been trying to deal with tank fungus and fish infections. This message from Audrey Halpert, at Albert Bridge School, provides an example of one such problem. I am having a massive die-off. Most seemed to be up and feeding well for two or three weeks then they started to get skinny and lie on the bottom of the basket. The dead and dying each have white patches that look like fungus on their operculum. We have lost 20 of our 70 and there are 10-15 more sick. I have tried to separate sick from healthy in separate baskets. Our water parameters are fine except the high Gh. What can I do? I don’t want to lose them all. Is this ick? By the way, Audrey's water chemistry numbers were all perfect. Here are a couple of pictures of Audrey's fish with their "white patches." I asked Audrey if the white stuff looked like Saprolegnia. She responded with: Joe, I wouldn’t say it's that cottony or fluffy, but maybe I should scrape some and look at it under a microscope. It definitely shows up on the operculum, not on other parts of the body yet. It seems to affect their breathing. I'm afraid I may lose them all. After she looked at it under a microscope, she also though it didn't look like ich, which two different experts on the TIC/SIC network suggested it might be. Stumped, I sent Audrey's photos to Dartmouth Professor Robb Cramer. He sent me this reply: Based on those 2 photos, it does not look like a fungus to me. It is hard to say what it is based on those 2 photos. The lack of feeding indicates something is wrong, but super hard to figure out the cause with limited information. There is some chance it is a disease called Columanaris, but just speculating. My global advice in these situations is to do a 50% water change ASAP. Clean the gravel with the siphon. Separate any sick fish and put them in another small tank if you can. Audrey came back with the photo below and this: It’s odd that the patches are always on the gills. I may remove the dying to a home tank in a cool workroom I think they are goners any way. Even though things weren't going well, and I hadn't been able to be helpful at all, I complimented Audrey on her photography skills. See these closeups (the second is taken at a later date). Robb recommended two treatments, first an API fish antibiotic and, if that didn't solve the problem, an API anti-fungal treatment. They look like this. After all Audrey's worrying and Robb and Audrey's efforts, it's not clear that her fish are going to make it. Most of these long-distance consultations aren't this challenging and work out better. By the way, when I complimented Audrey for the great closeup photos she was sending, she said that she took them using a ProScope attachment for her iPad. Here's what that $149 device looks like. Two-headed trout still with us! Mary Fiedler, of Cambridge Elementary School, sent this picture on 3/15/18 of her school's two-headed alevin. Still hanging in there! Too much suction! Jaclyn Bristol, of Guilford Central School, sent me this e-mail. We have a high number of fish dying this week. It looks like even with our filter end covered, they get sucked onto it and cannot get off, resulting in them dying. We have moved them back into the cribs for now. We are down to 77 fish. What are the rough die rates? I told Jaclyn that the problem was that her fish weren't yet strong enough to swim off the filter-end netting. Her decision to put the trout back into the breeder basket was a good one. That way they can get stronger and should be able to resist the suck of the filter in a week or two. Another option is to reduce the rate of flow of the filter. (We normally urge teachers to keep their filters at maximum flow, but this situation is an exception to that rule.) For those who have the AquaClear 110 filter, page 12 of your Instruction Manual contains this image, showing where the nob is to adjust flow rate. (It's that red thing poking up!) Most other filters will have similar ways to adjust flow rate.
Regarding Jaclyn's question about mortality rates, I answered that the 2016-2017 survival rate was 59% and that the rate last year (2017-2018) was 47%. Here is a ranked list of the principal causes of mortality during 2017-2018. The number at the end of each line is the number of schools (of the 21 completing a Survey Monkey questionnaire I sent them) indicating that that particular item was a principal cause of their fish losses.
This is why we worry so much about catching the swim-up stage. Send your swim-up data! We still need to hear from more schools.
0 Comments
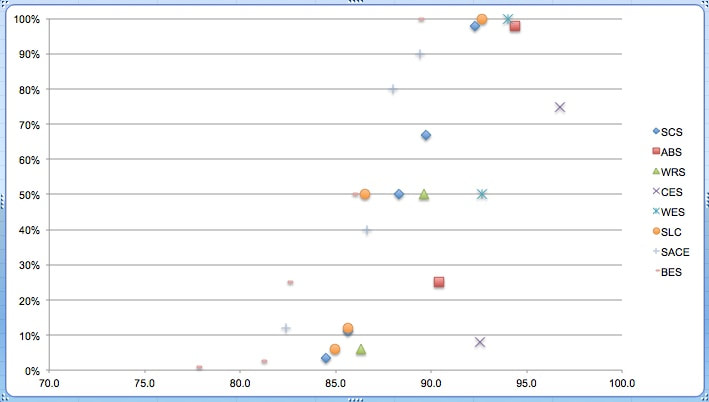
Students tell their schools about TIC! Kids work on improving habitat. F&W research. DI, DI, DI!3/13/2019 Student TIC presentation Suzanne Alfano, of Maple Street School, sent me a link to a great Google Slides presentation by her 5th grade students. Here's what Suzanne said in sending me the link. Here is a copy of the Google Slides presentation that the 5th graders gave at our all-school meeting. I gave the students the format for what they should include, and they did all the research and slide building. The close-ups that the one teacher is providing are unbelievable! (Way to go, Danielle!) We are collaborating with the history teacher and English teacher on a civics/letter writing/science lesson. Click the image above to go to the presentation by Suzanne's students. NH students work on habitat The current issue of the fine publication Forest Notes contains a great story about how students at Kearsarge Regional High School are partnering with the Forest Society to improve native brook trout habitat. Here's what the story looks like. Click on either of the page images below or on the name of the magazine (above) to access the story, which you'll find on pages 12 and 13. Research on the Batten Kill Vermont Fish and Wildlife fisheries biologist Lee Simard recently sent me several interesting reports on the Batten Kill, its trout population, and the work that has been done to improve trout habitat on what's known in southwestern Vermont as "the Kill." Depending on the age, reading ability, and interest of your students, these might make good "complex texts" for them to read. Whether or not it's appropriate reading material for your students, there's a lot to be learned about trout, trout habitat, and the kind of scientific research that career fisheries biologists engage in. Here are the four research reports. Using telemetry to track trout movement Click on the title above to access the study. The image below shows you what a telemetry transmitter looks like. Assessment of "cover" on Batten Kill Click on the title above to access this report, which studied not only the Batten Kill but four other Vermont rivers, some of which might be closer to you: the Castleton, Dog, Mettawee, and Poultney Rivers. Trout population response to cover Click on the title above to access the study. Here's a photo of the kind of in-stream "cover habitat" that can increase trout populations. Also, click on the image below to see the Fish and Wildlife Department's plan for management of the Batten Kill. (I think you can probably find other river management plans that the F&W Department has developed.) It's me again: What's the relationship between DI and swim-up? Every year, we want to refine our understanding of the relationship between DI, as we calculate it using our home-grown "Temp and DI record and swim-up calculator." We can't do that without input from you. Here's what some past data look like. As you can see, the first signs of swim-up occurred as early as a DI of 82, but several more schools began to notice swim-up behavior when their DI hit 85. Most fish had swam up when the DI was 93. But these generalizations are based on data from just eight schools, so I'd like to expand our pool of data by including your numbers.
Here's some of the 2019 data teachers have already submitted:
So what happened at your school? Please send in your data. Most helpful would be
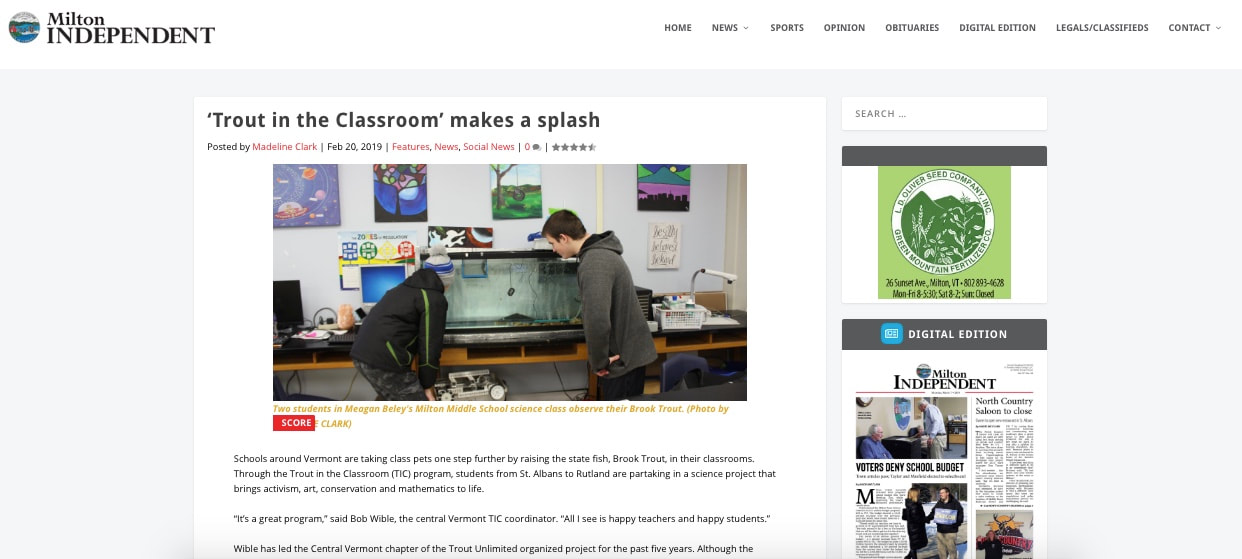
New images of trout. Impressive TIC interpretive dance! Good press! Temperature and bacteria.3/10/2019 Cool trout-themed designs Lisa Marks, of Ludlow Elementary School, is always looking for previously undiscovered TIC-related materials on the Internet and in her local stores. Often she finds things I never knew of--like this. Apparently this product line, which Lisa found in a store in Ludlow, also includes, bookmarks, tee-shirts, and refrigerator magnets. BVS TIC media Jeff Walker, new TIC teacher at Benson Village School, sent me some great examples of what his students are doing with TIC at his school. Here is a photo of posters they created. Even more unusual was the interpretive dance one of Jeff's students performed (below). Way to go Jeff and student! Anomalies Every year some if not most (all?) of our TIC tanks observe one or more forms of biological anomalies. Here's yet another sent in on February 8 by Mary Fiedler, of Cambridge Elementary School. Can you spot the two-headed alevin? (It's near the bottom of the image.) Schoolhouse Learning Center images Danielle Levine, of SchoolHouse Learning Center, sent several great images. Here's the caption she submitted with the following photo. Our trout are swimming up and feeding. And some have escaped through a little hole in our breeder basket. We are calling them Houdini. Based on the size and color of some of Danielle's fry, I'd guess that she and her students used the "warm and fast" temperature protocol. Good looking fish, huh? Here's a photo of the SLC tank with their escaped "Houdini fry." Danielle also sent me some close-up images of their beautiful fry. Thanks, Danielle. You do a service to us all by sharing these gorgeous close-up photos! Essex High School fry Kelly Hill, of Essex High School, sent this video of their fish. It's always great to see what the fry look like at other schools. Nice publicity for Milton MS TIC! Bob Wible told me about a great article that appeared in the Milton Independent. Some of the facts are a bit off, but it's great publicity for the program. Click the image below to read it. Temperature and bacteria A few schools are still keeping their tanks cold. We don't recommend that at this stage of the process. For one, the good bacteria we put in the tank, NovAqua Plus in our case, really don't grow at temperatures below 50. This means that they will not become those "biological machines" that
(You'll control nitrate levels by periodic partial water changes.) Secondly, at colder temperatures, you may find that your alevin/fry aren't likely to swim up. The swim-up instinct is correlated with a certain degree of development--a DI of approximately 85 by our method of calculation. But it's also tied to light and to water temperature. That's why at this point we urge you to raise your temperature to 52 degrees and to remove the top and front foam panels during the school day. Swim-up questions Unusually for me, I haven't written a new blog post in several weeks. Normally I try to post a new blog every five to seven days, but a trifecta of distractions--running for our town's select board, a two-week visit with my younger daughter's family in MD, and 10 days of a bad cold--cut into my time at the keyboard. I have, however, been hearing from and responding to TIC teachers, including questions related to swim-up. A fair number of teachers have reported that their fish have swum up and are eating. Yea! In several of those cases, teachers found that the swim-up occurred at or around a DI of 85. Yea, again! (That's what past experience has suggested might be the moment when swim-up would occur, but that conclusion is based on too little data, so we desperately want more data to modify or confirm our DI target. In a few cases, however, teachers whose DIs were in the neighborhood of 85 reported that they couldn't observe any swim-up activity. When I asked follow up questions, I learned that the water in those tanks was in the mid-40s, and the teachers still had the tank fully shrouded with foam. So, if your fish haven't swum up yet, do this:
Problems across TIC country Most days those of us on the national TIC/SIC e-mail network get several messages, either from teachers asking questions or from more experienced teachers and volunteers who think they might have answers to offer. Here are some representative exchanges. Cannibalism! On 2/18, teacher Michele Ferrel posted this message: My fish are eating each other and have been since the beginning. As of Friday I was down to 11 fish. Ten fish have been eaten in less than two weeks. I am trying to balance feeding them the right amount without creating too much waste to make the parameters go too high. I have been doing this for years and this is the first time I have had fish being eaten at this rate. Later that day, Maryland TIC coordinator Chuck Dinkel offered this response: When predation takes place in the tank, the big fish eat the little ones. Usually there are only a few big fish involved in the predation. One of the suggestions we make to teachers in Maryland who experience this problem is to net the big fish, put the breeder basket back in your tank, and give the big fish a "time out." One of the reasons that there can be a big size difference among the fish in your tank is that the smaller fish hang out near the bottom and don't get much of the food added to the tank because it tends to float awhile before sinking, The bigger fish get it first. The solution is to clear a quarter of your tank's bottom at one of the ends. Then fill a beaker with a small amount of tank water. Add fish food to this water and let it soak until it sinks. Suck it up with your turkey baster, point the baster towards the gravel-free portion of the tank and shoot the food to that area. The bottom-dwelling small fry will eat it and eventually get big enough that they don't become a meal for the big guys. You still need to put a small amount of food in the breeder basket for the big fish. Eventually the size difference will be minimized, and you can return the big fish back to the tank. Gross build-up in hoses On 2/19, Mary Soriano posted this (you may recalled that I described Mary as the TIC Godmother; after decades when she wasn't raising trout, she just rejoined the program this year): Hi there. Our levels are testing good but look at our piping. Is this normal? My chiller from 30 years ago was a canister chiller so I didn’t have piping like this. Anyone else experience this? Here are two photos Mary attached to her e-mail. One of the first responses came from "Stetson," of the Arizona TIC program, who wrote:
Those are the clumps of cells that make up the colonies of your nitrifying bacteria, which can be opaque white to brownish in color . What you see are actually clumps of bacteria stuck together in their own slim matrix or as we like to say the "bacterial biofilm". Some people "worry" and are "not happy" about the appearance. That is why some filter manufacturers use colored or opaque tubing for their filters. If you have a gram stain kit and a microscope, you may find it interesting to identify whether the bacteria are gram negative or gram positive and whether they are rod shaped or cocci. A brush and some diluted Chlorox bleach will clean the tubing at the end of the year, or you can go to your local hardware store and get new tubing to start with next year. Our Maryland friend Chuck Dinkel added this to the conversation: Mary, Following up on Stetson's hose cleaning advice. Make sure you clean the hoses as soon as the fish are released. If you allow the tubing to dry out, it becomes more difficult to clean. Get a bottle brush the fits the diameter of the tubing, remove the tubing from the chiller, run a stiff piece of wire (I use electrical wire) through the tubing and then attach the bottle brush. Bend the wire around the end of the brush. You can now pull the brush all the way through the tubing and clean the entire piece of tubing. This also works for any hoses that you may have for the filter. So you can see the benefits of our national TIC/SIC support system! |
Joe Mark is Lead Facilitator of Vermont's Trout in the Classroom program.In June 2012, I retired after 40 years in higher education, having spent the last 32 years of my career as dean at Castleton. One of the first things I volunteered to do in retirement was to work with a parent-friend to help the Dorset School, where his kids and my Vermont grandkids attend, start a TIC program. Gradually that commitment grew into my current role, which is both demanding and highly rewarding. Archives
December 2019
Categories |




















 RSS Feed
RSS Feed
